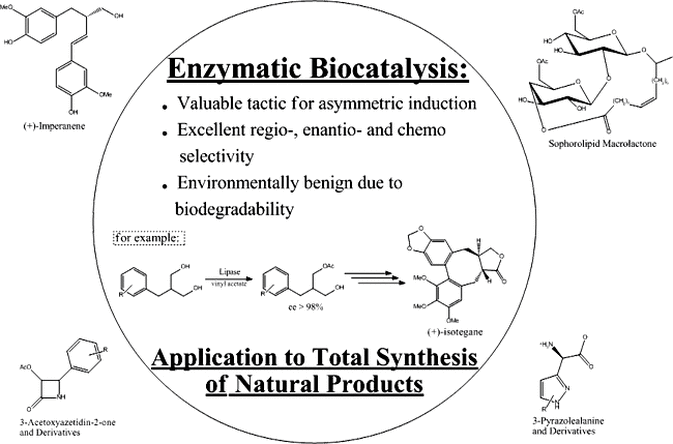
Research Interests I: Glycolipid Modification Using Enzymes, Namely Sophorolipids.
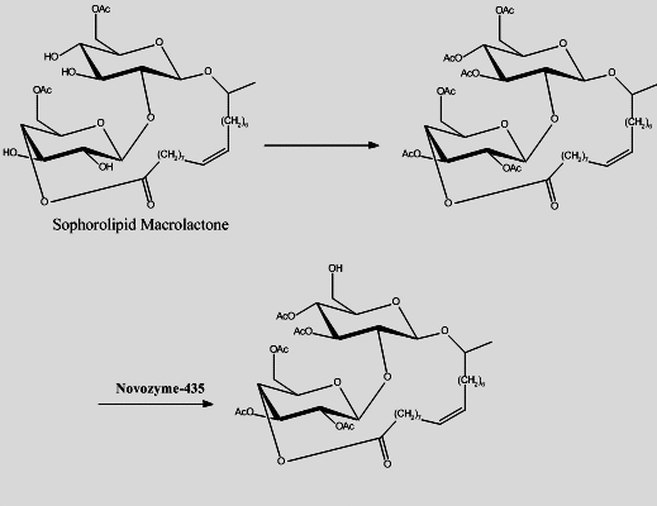
Lipopolysaccharides (LPS), specifically bacterial lipopolysaccharides, have been known to possess beneficial antitumor and immuno-pharmacological effects. However, LPS impose undesired effects or toxicity and the recent decade has seen an explosion in the synthesis of LPS tailored to produce desired pharmacological responses while minimizing the undesirable side effects. Because of the pharmacological benefits and low toxicity exhibited by complex glycolipids such as sophorolipids or rhamnolipids (derived from renewable agrochemicals) as immuno-modulators, we are currently synthesizing and evaluating well-defined glycolipid analogs using a variety of enzymatic and chemical steps in the synthesis where these respective approaches are most effective. The derivatives synthesized, thereof, will then be used to identify leading potential drug candidates.
Lipopolysaccharides (LPS), specifically bacterial lipopolysaccharides, have been known to possess beneficial antitumor and immuno-pharmacological effects. However, LPS impose undesired effects or toxicity and the recent decade has seen an explosion in the synthesis of LPS tailored to produce desired pharmacological responses while minimizing the undesirable side effects. Because of the pharmacological benefits and low toxicity exhibited by complex glycolipids such as sophorolipids or rhamnolipids (derived from renewable agrochemicals) as immuno-modulators, we are currently synthesizing and evaluating well-defined glycolipid analogs using a variety of enzymatic and chemical steps in the synthesis where these respective approaches are most effective. The derivatives synthesized, thereof, will then be used to identify leading potential drug candidates.
Research Interests II: Chemoenzymatic Synthesis of β-lactams and β-Sultams
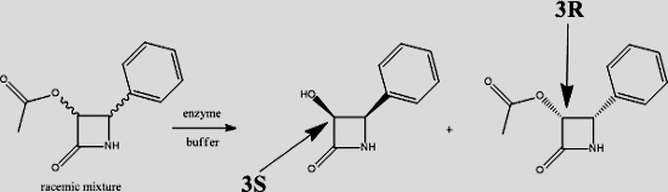
We are also pursing the development of new chemo-enzymatic methods for the synthesis of homochiral compounds of clinical significance with particular focus on using enzymes for stereocontrolled processes pertaining to resolution of racemates or chiral induction of prochiral substrates. One such focus is on the synthesis of optically active cyclic lactams that are versatile building blocks in organic synthesis (ε-caprolactams) and a very important class of antibiotic, the β-lactams.
We are also pursing the development of new chemo-enzymatic methods for the synthesis of homochiral compounds of clinical significance with particular focus on using enzymes for stereocontrolled processes pertaining to resolution of racemates or chiral induction of prochiral substrates. One such focus is on the synthesis of optically active cyclic lactams that are versatile building blocks in organic synthesis (ε-caprolactams) and a very important class of antibiotic, the β-lactams.
Research Interests III: Chemoenzymatic Synthesis of Small Natural Products
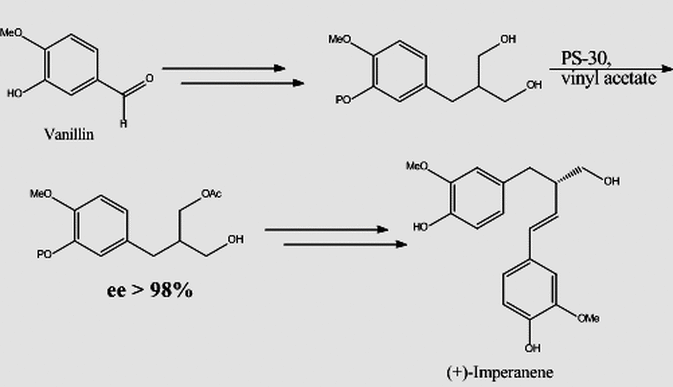
One aspect of our research deals with the use of enzymes for the synthesis of chiral synthons as precursors to molecules of biological importance. Enzyme-catalyzed reactions provide us the opportunity for high levels of structural control because appropriate selection of right enzyme allows us to control the regio-, chemo- and stereoselectivity of certain chemical reactions that are difficult or impossible to achieve by way of conventional chemical synthesis. In addition, these reactions occur under mild conditions, hence functionally sensitive molecules can be manipulated under ambient or near ambient conditions and in many cases targeted reactions may be realized without the need for necessary protection/ deprotection steps. In the synthesis of imperanene, a natural anticoagulant, the enzyme PS-30 (from Psuedomonas cepecia) was used in converting a prochiral 1,3-diol into a highly enantiomerically enriched chiral intermediary toward the total synthesis of the desired compound.
One aspect of our research deals with the use of enzymes for the synthesis of chiral synthons as precursors to molecules of biological importance. Enzyme-catalyzed reactions provide us the opportunity for high levels of structural control because appropriate selection of right enzyme allows us to control the regio-, chemo- and stereoselectivity of certain chemical reactions that are difficult or impossible to achieve by way of conventional chemical synthesis. In addition, these reactions occur under mild conditions, hence functionally sensitive molecules can be manipulated under ambient or near ambient conditions and in many cases targeted reactions may be realized without the need for necessary protection/ deprotection steps. In the synthesis of imperanene, a natural anticoagulant, the enzyme PS-30 (from Psuedomonas cepecia) was used in converting a prochiral 1,3-diol into a highly enantiomerically enriched chiral intermediary toward the total synthesis of the desired compound.
Research Interests IV: Chemoenzymatic Synthesis of Non-Natural Amino Acids
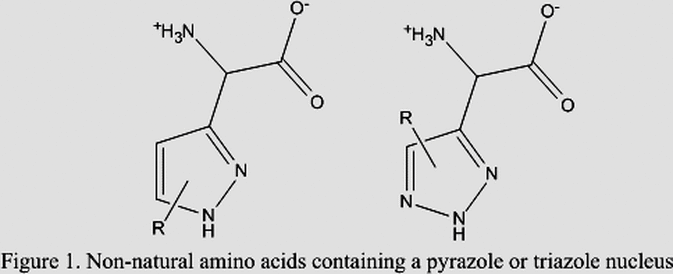
Our lab also focuses on the synthesis and biological evaluation of non-natural amino acids. The incorporation of non-natural amino acids into polypeptide chains has been shown to improve their biological and pharmacological properties, such as improved therapeutic half-life or modulated immunogenicity. Our synthesis of these amino acids is attractive because we start from relatively cheap and abundantly available starting materials and once again we utilize enzymes in these syntheses for chiral resolution of the racemates thus synthesized. This provides efficient methods for the synthesis of enantio-enriched non-natural amino acids, specifically those containing pyrazole or triazole nuclei. β-(pyrazole-1-yl)-L-alanine, a non-protein amino acid, which is found in watermelon seeds serves as the template for which the syntheses are constructed.
Our lab also focuses on the synthesis and biological evaluation of non-natural amino acids. The incorporation of non-natural amino acids into polypeptide chains has been shown to improve their biological and pharmacological properties, such as improved therapeutic half-life or modulated immunogenicity. Our synthesis of these amino acids is attractive because we start from relatively cheap and abundantly available starting materials and once again we utilize enzymes in these syntheses for chiral resolution of the racemates thus synthesized. This provides efficient methods for the synthesis of enantio-enriched non-natural amino acids, specifically those containing pyrazole or triazole nuclei. β-(pyrazole-1-yl)-L-alanine, a non-protein amino acid, which is found in watermelon seeds serves as the template for which the syntheses are constructed.